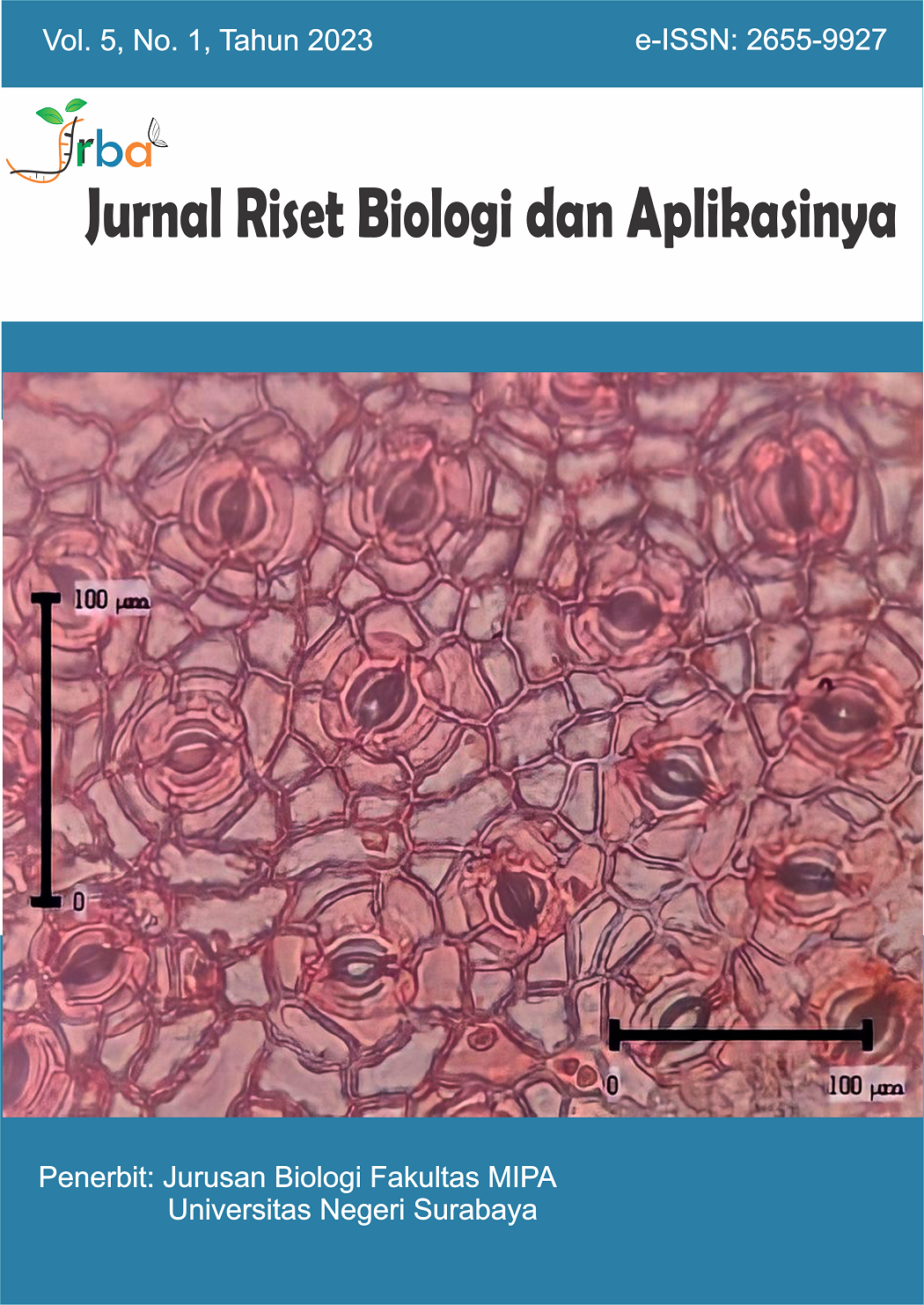
Leaf and Stomata Morphometrics of Gayam Inocarpus fagifer (Fabaceae) at Different Altitudes
DOI:
https://doi.org/10.26740/jrba.v5n1.p16-26Abstract
Gayam (Inocarpus fagifer) is one of the members of the angiosperm flora in Ambon City, Indonesia, that grows and develops at various altitudes. This research aimed to analyze the leaf and stomata morphometrics of these plants in the Aer Louw and Ema Village areas. Leaf samples were taken from the upper, middle, and lower strata and considered as replicates. The morphometric characteristics were measured manually using millimeter block paper and the formula for calculating leaf ratio. Furthermore, the stomata were stained using the direct incision method and safranin. The incision results were analyzed using an Olympus CX23 microscope at 400x magnification. The measurement and observation were analyzed descriptively and correlatively. The results showed that the average leaf width and length, also the midrib length were greater in Aer Louw Village than in Ema Village; while the leaf tip and stalk length were greater in Ema Village than in Aer Louw Village. The characteristics of stomata length and width in Ema Village were greater than in Aer Louw Village; otherwise, the number, index, and density of stomata in Aer Louw Village were greater than in Ema Village. Meanwhile, the correlational analysis showed that the environment influenced the variations of leaves and stomata. Therefore, the variations of leaves and stomata in the areas could predict plant adaptations to different environments.
References
Elhaddad, N. S., Hunt, L., Sloan, J., & Gray, J. E. (2014). Light-induced stomatal opening is affected by the guard cell protein kinase APK1b. PLoS One, 9(5), e97161.https://doi.org/10.1371/journal.pone.0097161
Fritz, M. A., Rosa, S., & Sicard, A. (2018). Mechanisms underlying the environmentally induced plasticity of leaf morphology. Frontiers in Genetics, 9, 478. https://doi.org/10.3389/fgene.2018.00478.
Gao, J., Song, Z., & Liu, Y. (2019). Response mechanisms of leaf nutrients of endangered plant (Acer catalpifolium) to environmental factors varied at different growth stages. Global Ecology and Conservation, 17, e00521. https://doi.org/10.1016/j.gecco.2019.e00521
Haworth, M., Marino, G., Materassi, A., Raschi, A., Scutt, C. P., & Centritto, M. (2022). The functional significance of the stomatal size to density relationship: Interaction with atmospheric [CO2] and role in plant physiological behaviour. Science of The Total Environment, 863, 160908. http://dx.doi.org/10.1016/j.scitotenv.2022.160908.
He, J., & Liang, Y-K. (2018). Stomata. Plant Science. https://doi.org/10.1002/9780470015902.a0026526.
Hong, T., Lin, H., & He, D. (2018). Characteristics and correlations of leaf stomata in different Aleurites montana provenances. PLoS One, 13(12), e0208899. https://doi.org/10.1371/journal.pone.0208899
Kumari, K., & Yadav, S. (2018). Linear regression analysis study. Journal of the practice of Cardiovascular Sciences, 4(1), 33.
Li, X., Li, Y., Zhang, Z., & Li, X. (2015). Influences of environmental factors on leaf morphology of Chinese jujubes. PLoS One, 10(5), e0127825. https://doi.org/10.1371/journal.pone.0127825
Paembonan, S. A., Larekeng, S. H., & Millang, S. (2021). The dynamics of physiological properties of ebony (Diospyros celebica Bakh.) based on crown position and altitude. Earth and Environmental Science, 807. https://doi.org/10.1088/1755-1315/807/3/032016
Sun, J., Liu, C., Hou, J., & He, N. (2021). Spatial variation of stomatal morphological traits in grassland plants of the Loess Plateau. Ecological Indicators, 128, 107857. https://doi.org/10.1016/j.ecolind.2021.107857
Turkheimer, E., & Waldron, M. (2000). Nonshared environment: a theoretical, methodological, and quantitative review. Psychological Bulletin, 126(1), 78. https://doi.org/10.1037/0033-2909.126.1.78
Wang, C., Lu, H., Zhang, J., Mao, L., & Ge, Y. (2019). Bulliform phytolith size of rice and its correlation with hydrothermal environment: A preliminary morphological study on species in Southern China. Frontiers in Plant Science, 10, 1037. https://doi.org/10.3389/fpls.2019.01037
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2023 Jurnal Riset Biologi dan Aplikasinya

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
 Abstract views: 840
,
Abstract views: 840
, PDF Downloads: 413
PDF Downloads: 413